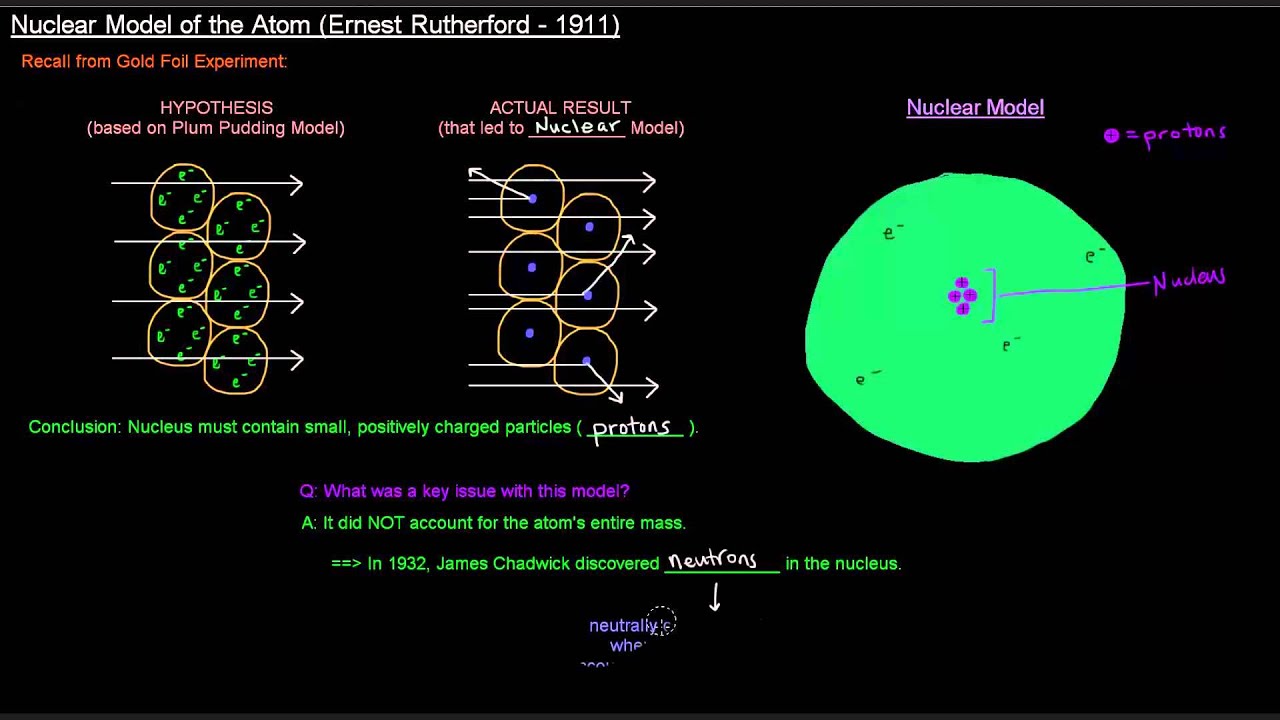
It does not explain the location of electrons in an atom. It does not explain the stability of atoms.ģ. It does not explain the spectrum of light emitted by atoms.Ģ. Atomic Model (Atomic Theory) - An atom is the smallest unit of ordinary matter that forms a chemical element.Atoms are made of fundamental particles called protons, neutrons and electrons. The Rutherford atomic model has some limitations.ġ. Rutherford’s experiment showed that most of the atom was empty space, with the nucleus at the center. Thomson’s model of the atom consisted of a small, positively charged nucleus surrounded by negatively charged electrons. Rutherford used a gold foil to test the model of the atom proposed by J.J. The Rutherford model experiment was an experiment conducted by Ernest Rutherford in 1911. It also showed that the nucleus has a powerful force that can deflect the alpha particles. This experiment showed that the nucleus is very small compared to the size of the atom. However, many of the alpha particles were deflected (changed direction) by the atoms. They expected that the alpha particles would pass right through the gold atoms. To test this model, Rutherford and his team fired tiny particles called alpha particles at a thin sheet of gold. His model is based on the idea that the atom is mostly empty space with a small, dense nucleus at its center. In 1909, physicist Ernest Rutherford proposed a model for the atom that is still used today. Experiment with the Rutherford Atomic Model It was also able to explain the stability of the atom. The Rutherford model was able to explain the results of the alpha particle experiments. The electrons orbit the nucleus in shells. The nucleus is held together by the strong nuclear force. In Rutherford’s model, the atom is a small, dense, positively charged nucleus surrounded by a cloud of negatively charged electrons. Rutherford’s model was based on his experiments with alpha particles. Rutherford’s atomic model became known as the nuclear model. The nucleus is the tiny, dense, central core of the atom and is composed of protons and neutrons. Rutherford’s model was based on the Plum Pudding model which was developed by J.J. He concluded that all of the positive charge and the majority of the mass of the atom must be concentrated in a very small space in the atom’s interior, which he called the nucleus. The Rutherford atomic model is a model of the atom that was developed by Ernest Rutherford. This indicated that the gold atom was not uniform, but had a small, dense nucleus. He observed that some of the alpha particles were deflected by the foil. In this experiment, Rutherford fired a beam of alpha particles at a piece of gold foil. The Rutherford model was based on the results of Rutherford’s gold foil experiment. The nucleus is held together by the strong nuclear force, and the electrons orbit the nucleus in shells. In this model, the atom is a small, dense, positively charged nucleus surrounded by electrons. Rutherford atomic structure is a model of the atom that is based on the work of Ernest Rutherford. He concluded that the atom must be composed of a small, dense nucleus surrounded by electrons. He observed that some of the alpha particles were deflected, suggesting that they were being scattered by something in the atom. The Rutherford nuclear model was developed after Rutherford performed an experiment in which he fired a beam of alpha particles at a piece of gold foil. The nucleus is composed of protons and neutrons, and the electrons orbit around the nucleus in shells. The model is based on the idea that the atom is composed of a small, dense nucleus surrounded by electrons. The Rutherford nuclear model is a model of the atom that was developed by Ernest Rutherford in 1911. 
The scattering of alpha particles by atoms. The emission and absorption of light by atoms. The relatively small size of the atom compared to the size of the nucleus. The Rutherford model was able to explain the following features of the atom: The model was able to explain several features of the atom that were not explained by the earlier model of the atom, the plum pudding model. 
The model is based on the idea that the atom is mostly empty space, with a small, dense nucleus in the center. 
AND if they already knew that the electron was small and negative, then the atom must have a small positive nucleus with the electrons around them.Rutherford’s atomic model is a model of the atom that was proposed by Ernest Rutherford in 1911. If the positive alpha particles mostly passed through the foil, but some bounced back. How could that be if the plumb pudding model was correct? Rutherford's experiment prompted a change in the atomic model. Rutherford found that most of them went right through the foil. If you shoot these positive alpha particles at this positive pudding atom, they should mostly bounce off, right? Well, that is not what happened. He shot some alpha particles (which are really just the nucleus of a helium atom) at some really thin gold foil. Ernest Rutherford said one day "hey, I think I will shoot some stuff at atoms." I am sure his wife said "oh, Ernie" (she probably called him Ernie) "if it makes you happy to play with your little physics stuff, go ahead.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
